Pocket K No. 62: Delivering Durable Genetic Resistance in Corn
| |
Introduction
Corn is the world's most-produced grain. In the US, the top producer of corn, the production volume for 2024/2025 was estimated at 377.6 million metric tons, with 2024 exports reaching 62.5 million metric tons. Trailing the US as the top producer, China and Brazil are also significant global corn producers, ranking second and third, respectively.1,2
In Sub-Saharan Africa, the well-being of millions of smallholder farmers and consumers is intrinsically linked to corn (maize) production, which is fundamental for food security, poverty alleviation, health, economic progress, and stability. With global corn demand expected to surge by 2050 due to population growth,3 the vulnerability of corn harvests to biotic and abiotic threats underscores the urgent need to develop more resilient corn varieties.4
A Major Biotic Threat to Corn
Pathogenic fungi impact corn production by causing rot in the ears and stalks leading to lodging and yield losses. These ear and stalk rots are caused by several fungi, including Fusarium and Aspergillus (Figure 1).
Fusarium and Aspergillus not only reduce yields, but they also destroy grain quality by producing mycotoxins. Mycotoxins are poisonous chemicals that cause severe illness and death in humans and animals. The most notorious mycotoxins are the highly potent aflatoxins from Aspergillus, and the increasingly common vomitoxin (fumonisins) and other toxins from Fusarium.
In the US, contamination of corn by mycotoxins results in large economic losses due to lost yields and contaminated grain. The Crop Protection Network documented 3.6 million tons of corn harvest lost in 2018 to Fusarium stalk and ear rot. Beyond these direct yield reductions, substantial costs are incurred by producers, processors, and marketers to comply with regulations designed to minimize human exposure, for example the use of binders. Further economic losses for producers occur when contaminated grains are bought at reduced prices or rejected completely. The combined economic impact of mycotoxins on food and feed in the US has been estimated at US$2.8 billion annually.5,6
Beyond economic losses, mycotoxins also pose a significant public health threat, with more than half the world’s population—an estimated 4.5 billion people—exposed to deadly aflatoxins every year. Mycotoxins occur more frequently under tropical conditions, and people in developing countries have high exposure and limited mitigation options. A stark example occurred in Kenya in 2004, when suspected aflatoxin contamination in corn led to 317 reported illnesses and 125 deaths across seven districts, prompting an official outbreak declaration by both Kenyan authorities and the World Health Organization. Moreover, aflatoxin contamination has been linked to a substantial number of liver cancer cases in Africa, Southeast Asia, and the Western Pacific.7,8,9
Extreme climate conditions and other abiotic stresses are intensifying fungal infections and the associated problem of mycotoxins. For example, Italy experienced unusually dry and hot summers in 2003, 2004, and 2012, which favored the proliferation of Aspergillus flavus. The proliferation led to its dominance over Fusarium spp. in corn and a subsequent sharp rise in dangerous aflatoxin levels. Looking ahead, trajectories for 2031-2040 in 15 US states encompassing the Corn Belt project that nearly 90% of corn-growing counties will develop ideal conditions for Aspergillus infection and lead to increased aflatoxin contamination.10,11,12
Next-Generation Breeding to Combat Fungal Pathogens
The economic and health impacts of plant fungi and their mycotoxins can be minimized by developing corn varieties with durable resistance to fungal infection.
Resistance to Fusarium and Aspergillus species has been a vexing challenge to corn breeders due to the complicated biology of these fungi. Aspergillus flavus has a complex life cycle, capable of surviving on both living and dead plant material. Fusarium graminearum, on the other hand, initially lives inside living plant cells without killing them, before transitioning to a necrotrophic phase where it kills and consumes the plant tissue.13 Because these toxin-producing fungi can infect corn while it is growing and also survive in the harvested grain throughout storage and processing phases, a very important goal is to boost the corn’s immune system to recognize these intruders at early stages of infection.
Understanding Plant Immunity
Plants defend themselves against disease through an immune system that detects pathogens and triggers defenses to prevent them from entering plant cells and/or spreading through plant tissues. Detection occurs through a sophisticated surveillance system at the cell surface that detect specific clues to pathogen presence known as danger-associated molecular patterns (DAMPs) and pathogen-associated molecular patterns (PAMPs). These “patterns” are made up of fragments of the invaders, host tissue damage, and other signs of cellular stress, and they are recognized by sensors on the cell surface called pattern—recognition receptors (PRRs). PRRs recognize patterns that are common across different microbes, and therefore PRRs can often protect against a range of related pathogens.14
Not all plants have PRRs that recognize all pathogens, and even if plants do have the right PRRs some pathogens can evade or suppress detection and cause disease. However, scientists have found a way to fight back by showing that PRRs from one plant can be introduced into a susceptible plant and successfully enable recognition of and resistance to novel pathogens. 2Blades has supported efforts that enabled researchers to transfer a PRR called EF-Tu Receptor (EFR) from one plant family to another, conferring broad resistance in tomato plants to a range of bacteria. Other researchers have since shown EFR can protect wheat, oranges, apples, potatoes, and other crops from serious bacterial diseases. In another example, a tomato gene (Ve-1) conferred resistance to fungi in tobacco and cotton. 14,15,16,17,18,19
Plants also have a second major branch of their immune systems that scans for intruders inside cells. Intracellular receptors known as Nucleotide-binding Leucine-rich Repeat proteins (NLRs) detect “effector” proteins that the pathogen uses to suppress plant defenses. NLRs have exquisite sensitivity and mount a strong defense response that blocks disease progression. These responses are highly specific to certain pathogens or even particular races of pathogens that have the specific effector. For example, the barley (Hordeum vulgare) NLR gene Mildew locus A (Mla7) recognizes the effector AVRa7 from Blumeria hordei (Bh), triggering immunity against isolates carrying this effector. 20
2Blades has assembled a suite of gene discovery tools based on PRR, NLR, and other immune receptors to create its BladeSeek™ platform. 2Blades has used the platform to accumulate data for a range of crops and pathogens. The resulting comprehensive toolkit encompasses a large and growing gene anthology, rich phenotypic data, and powerful AI tools, that enables 2Blades’ researchers to rapidly identify and validate disease resistance genes in cereals, legumes, potatoes, and other key crops. This “digital” gene discovery and validation bring unprecedented speed and scale that complements traditional “analog” methods like chemical crop protection and conventional breeding.
2Blades is using the BladeSeek™ platform to analyze thousands of PRRs and NLRs in search of receptors that recognize Aspergillus and Fusarium. As these receptors are identified, the genes that encode them are introduced into high performing corn varieties to protect them from fungal attack and mycotoxin production.21
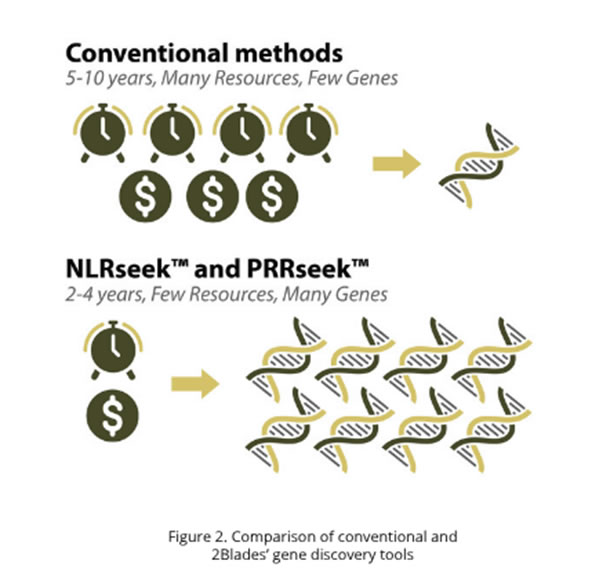
Using the BladeSeek™ tool for NLRs (NLRseek™) 2Blades has identified unprecedented numbers of resistance genes for the worst diseases of wheat: stem rust (Puccinia graminis f. sp. tritici), stripe rust (P. striiformis f. sp. tritici), leaf rust (P. triticina), wheat blast (Magnaporthe oryzae pathotype Triticum), and Fusarium head blight (F. graminearum). Moreover BladeSeek™ shortened gene discovery from an average of 5-10 years to only 2-4 years, discovering more genes with fewer resources (Figure 2).
Impact of Fungal-Resistant Corn Varieties
Durable resistance in corn against mycotoxigenic fungi offers a powerful solution for economic losses and enhanced well-being. By safeguarding yields and grain quality, the income of corn growers, handlers, and processors will also increase. Furthermore, reduced mycotoxin contamination translates to significant savings in mitigation costs and, crucially, ensures safer food and healthier lives for people, livestock, and aquaculture.
Upon the completion of development of a fungal-resistant corn variety, 2Blades will ensure successful deployment in the US through seed company partnering. 2Blades’ vision extends globally to transfer resistance genes, through strategic partnerships, into vital African tropical corn varieties, to protect people and livestock from mycotoxin contamination. This dual market approach is a core strategy at 2Blades and serves as a blueprint for future applications against Fusarium in wheat and barley.22
This Pocket K is produced by ISAAA Global Knowledge Center on Biotechnology and 2Blades. (April 2026)
References
- Shahbandeh, M. (2025, January 22). Corn production worldwide 2024/2025, by country. Statista. https://www.statista.com/statistics/254292/global-corn-production-by-country/
- U.S. Department of Agriculture Foreign Agricultural Services. (2024). U.S. Corn Exports in 2024. https://www.fas.usda.gov/data/commodities/corn
- United Nations. (2019). Population. https://www.un.org/en/global-issues/population
- Ekpa, O., Palacios-Rojas, N., Kruseman, G., Fogliano, V., & Linnemann, A. R. (2018). Sub-Saharan African maize-based foods: technological perspectives to increase the food and nutrition security impacts of maize breeding programmes. Global Food Security, 17, 48-56.
- Crop Protection Network. (2018, December 31). Corn Disease Loss Estimates From the United States and Ontario, Canada — 2018. https://cropprotectionnetwork.org/publications/corn-disease-loss-estimates-2018
- Robens, J. and Cardwell, K. (2003). The Costs of Mycotoxin Management to the USA: Management of Aflatoxins in the United States. Journal of Toxicology: Toxin Reviews, 22, 139-152. http://dx.doi.org/10.1081/TXR-120024089
- Williams, J.H., Phillips, T.D., Jolly, P., Styles, J.K., Jolly, C.M., and Aggarwal, D. (2004). Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. American Journal of Clinical Nutrition, 80, 1106-1122.
- Centers for Disease Control and Prevention. (2004, September 3). Outbreak of aflatoxin poisoning—eastern and central provinces, Kenya, January–July, 2004. Morbidity and Mortality Weekly Report 53(34);790-793. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5334a4.htm
- Liu, Y., & Wu, F. (2010). Global burden of aflatoxin-induced hepatocellular carcinoma: a risk assessment. Environmental Health Perspectives, 118(6), 818-824.
- Intergovermental Panel on Climate Change. (2022). Climate Change 2022: Impacts, Adaptation and Vulnerability. https://www.ipcc.ch/report/ar6/wg2/downloads/report/IPCC_AR6_WGII_SummaryVolume.pdf
- Paterson, R. R. M., & Lima, N. (2017). Thermophilic fungi to dominate aflatoxigenic/mycotoxigenic fungi on food under global warming. International Journal of Environmental Research and Public Health, 14(2), 199. https://doi.org/10.3390/ijerph14020199
- Yu, J., Hennessy, D. A., Tack, J., & Wu, F. (2022). Climate change will increase aflatoxin presence in US Corn. Environmental Research Letters, 17(5), 054017.
- Fountain, J. C., Scully, B. T., Ni, X., Kemerait, R. C., Lee, R. D., Chen, Z. Y., & Guo, B. (2014). Environmental influences on maize-Aspergillus flavus interactions and aflatoxin production. Frontiers in Microbiology, 5, 40.
- Lacombe, S., Rougon-Cardoso, A., Sherwood, E., Peeters, N., Dahlbeck, D., Van Esse, H. P., ... & Zipfel, C. (2010). Interfamily transfer of a plant pattern-recognition receptor confers broad-spectrum bacterial resistance. Nature Biotechnology, 28(4), 365-369.
- Schoonbeek, H. J., Wang, H. H., Stefanato, F. L., Craze, M., Bowden, S., Wallington, E., ... & Ridout, C. J. (2015). Arabidopsis EF‐Tu receptor enhances bacterial disease resistance in transgenic wheat. New Phytologist, 206(2), 606-613.
- Mitre, L. K., Teixeira-Silva, N. S., Rybak, K., Magalhães, D. M., de Souza-Neto, R. R., Robatzek, S., Zipfel, C., and de Souza, A. A. (2021). The Arabidopsis immune receptor EFR increases resistance to the bacterial pathogens Xanthomonas and Xylella in transgenic sweet orange. Plant Biotechnology Journal. 19:1294-1296.
- Piazza, S., Campa, M., Pompili, V., Costa, L. D., Salvagnin, U., Nekrasov, V., ... & Malnoy, M. (2021). The Arabidopsis pattern recognition receptor EFR enhances fire blight resistance in apple. Horticulture Research, 8.
- Boschi, F., Schvartzman, C., Murchio, S., Ferreira, V., Siri, M. I., Galván, G. A., ... & Dalla-Rizza, M. (2017). Enhanced bacterial wilt resistance in potato through expression of Arabidopsis EFR and introgression of quantitative resistance from Solanum commersonii. Frontiers in Plant Science, 8, 1642.
- Song, Y., Liu, L., Wang, Y., Valkenburg, D. J., Zhang, X., Zhu, L., & Thomma, B. P. (2018). Transfer of tomato immune receptor Ve1 confers Ave1‐dependent Verticillium resistance in tobacco and cotton. Plant Biotechnology Journal, 16(2), 638-648.
- Brabham, H. J., Hernández-Pinzón, I., Yanagihara, C., Ishikawa, N., Komori, T., Matny, O. N., Hubbard, A., Witek, K., Feist, A., Numazawa, H., Green, P., Dreiseitl, A., Takemori, N., Komari, T., Freedman, R. P., Steffenson, B., van Esse, H. P., & Moscou, M. J. (2025). Discovery of functional NLRs using expression level, high-throughput transformation and large-scale phenotyping. Nature Plants, 11(10), 2100–2114. https://doi.org/10.1038/s41477-025-02110-w.
- 2Blades. (n.d.). Gene discovery. https://www.2blades.org/our-activities/nlrseek
- Foundation for Food & Agriculture Research. (n.d.). Developing Durable Resistance to Fungal Threats in Corn. https://foundationfar.org/grants-funding/grants/developing-durable-resistance-to-fungal-threats-in-corn/